2024-03-21
On March 14, 2024, the U.S. Meals and Drug Administration accepted resmetirom (Rezdiffra)—the primary drug for a diabetes-related liver illness often called MASH.
The approval makes means for the primary and solely remedy for the remedy of a diabetes-related liver illness often called non-cirrhotic non-alcoholic steatohepatitis—often known as metabolic dysfunction–related steatohepatitis (MASH)—with reasonable to superior liver fibrosis (thickening/scarring).
“Beforehand, sufferers with [MASH] who even have notable liver scarring didn’t have a drugs that would instantly tackle their liver injury,” stated Nikolay Nikolov, MD, appearing director of the Workplace of Immunology and Irritation within the FDA’s Heart for Drug Analysis and Analysis. “ … approval of Rezdiffra will, for the primary time, present a remedy possibility for these sufferers.. .”
What’s Rezdiffra?
Manufactured by Madrigal Prescription drugs, Rezdiffra is an oral thyroid hormone receptor beta-selective agonist.
Rezdiffra is indicated for once-daily use. It’s for use together with food regimen and train.
The advisable dosage of Rezdiffra relies on precise physique weight.
For sufferers weighing lower than 220 kilos, the advisable dosage is 80 mg orally as soon as each day.
For sufferers weighing greater than 220 kilos, the advisable dosage is 100 mg orally as soon as each day.
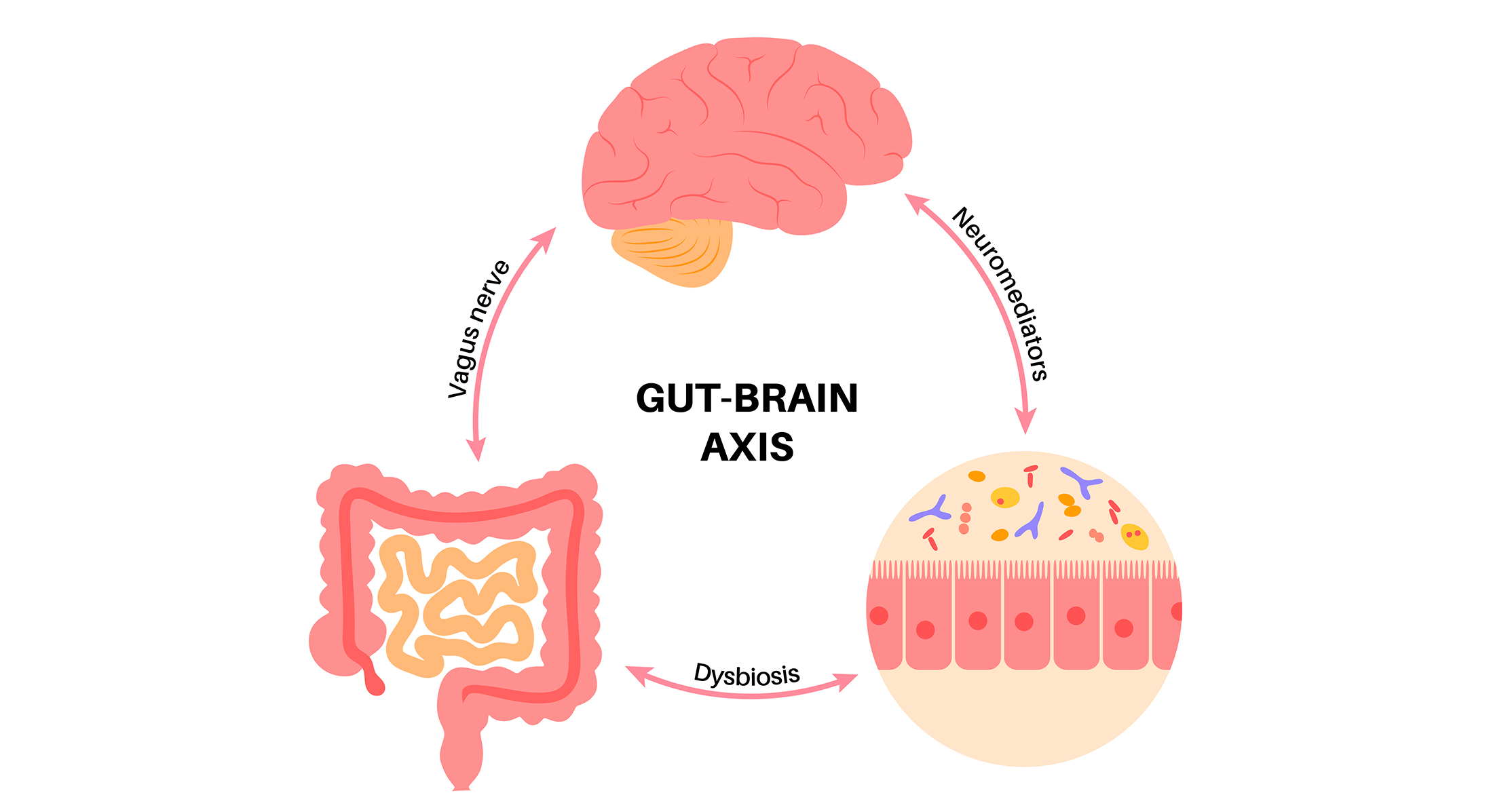
What’s MASH?
How Does Rezdiffra Assist Individuals With MASH?
In medical trials—to folks with biopsy-confirmed MASH—Rezdiffra was given, as soon as each day, at both 80-mg or 100-mg doses.
Following 52 weeks of remedy, each doses of Rezdiffra demonstrated statistically vital enchancment—in comparison with placebo—on two major endpoints:
MASH decision—together with a discount within the NAFLD exercise rating by ≥2 level) with no worsening of fibrosis, and
An enchancment in fibrosis by a minimum of one stage with no worsening of the NAFLD exercise rating.
What Are the Facet Impact of Taking Rezdiffra?
Rezdiffra shouldn’t be utilized in sufferers with an acute deterioration in liver operate.
The most typical hostile reactions reported in sufferers handled with Rezdiffra included diarrhea, nausea, pruritis, stomach ache, vomiting, constipation, and dizziness.
Use of Rezdiffra must be prevented in sufferers with deterioration in liver operate with cirrhosis .
Sufferers ought to cease utilizing Rezdiffra in the event that they develop indicators or signs of worsening liver operate.
When Will Rezdiffra Be Out there?
Rezdiffra is anticipated to be out there to sufferers in the US in April 2024, and shall be distributed by a restricted specialty pharmacy community.
Full prescribing data is accessible right here.