Ferring Prescription drugs has had editorial planning involvement, together with options of content material and subjects, in addition to proposal of specialists for articles. The ultimate academic content material was independently developed and accepted by the GMFH publishing crew and editorial board.
That is the sixth article in a particular assortment that focuses on the epidemiology, analysis and administration of C. difficile an infection, which is of relevance to gastroenterologists. See the primary article on C. difficile an infection prevalence and danger elements right here, the second article on diagnosing and managing major and recurrent C. difficile an infection right here, the third article on the professionals and cons of fecal transplants and outlined commensal consortia for recurrent C. difficile an infection right here, the fourth article on C. difficile an infection from clinicians’ and sufferers’ views right here and the fifth article on C. difficile issues right here.
The evolution of fecal microbiota transplants, one of the crucial superior microbiome-based remedies
Whereas fecal microbiota transplants (FMTs) have been practiced because the Fifties, it was not till just lately that they had been re-discovered as a therapeutic strategy for restoring an altered intestine microbiota composition and performance. Present European and American tips suggest FMTs for treating sufferers with recurrent Clostridioides difficile infections.
Initially, FMT was carried out after little processing of the unique fecal materials. But as key microbes or capabilities concerned in its efficacy have been characterised, firms have developed merchandise consisting of outlined consortia of microorganisms (particularly investigational microbiome biotherapeutics).
The approval of different therapeutic candidates for the microbiome can also be anticipated in early 2023 and can open the door to future biologics license purposes (a proper documentation required by the FDA to commercialize a brand new drug).
Challenges that hinder fecal microbiota transplants’ entry to market
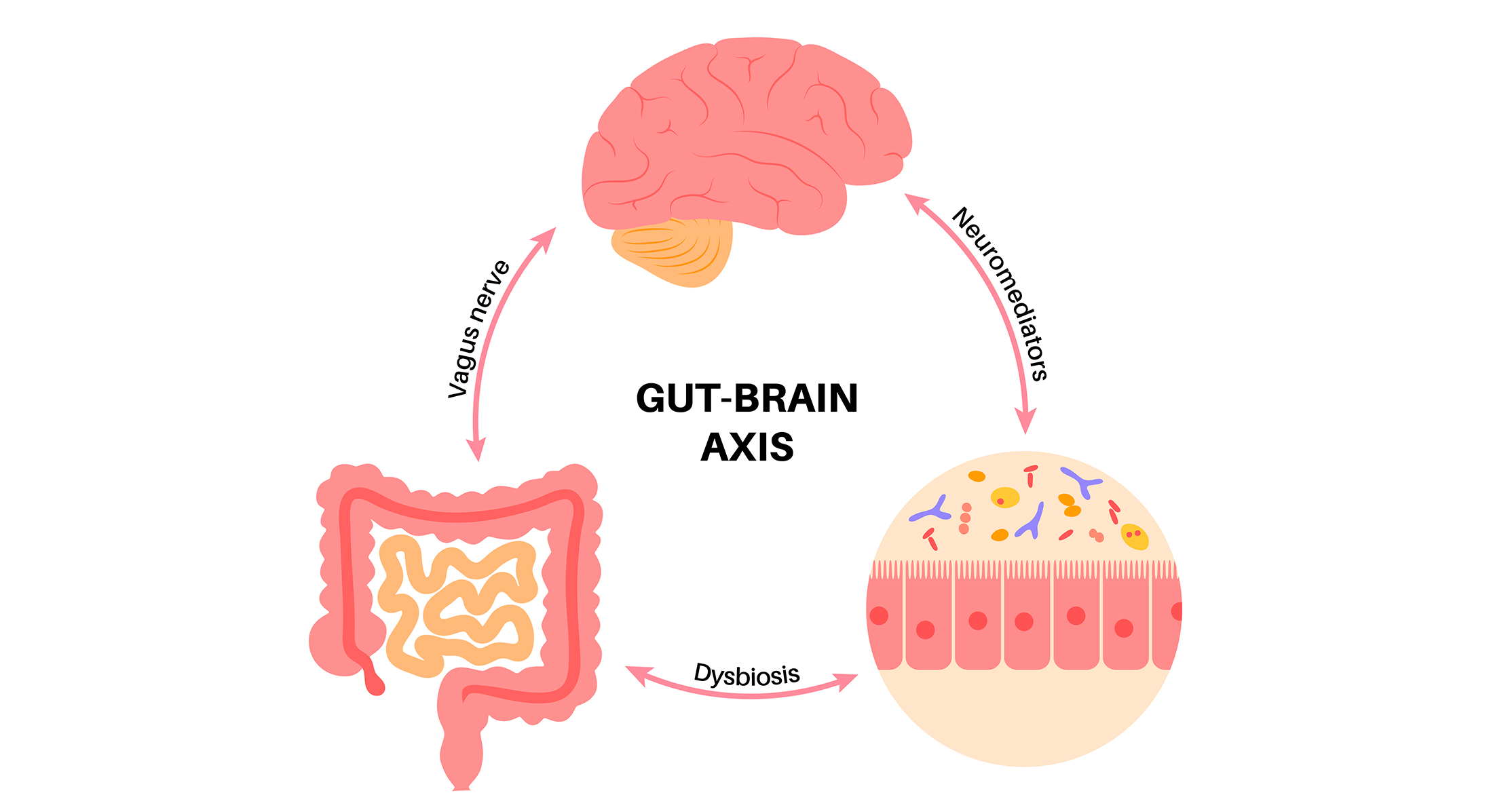
Whereas establishing stool banks has enabled a centralization of ready-to-use feces suspensions to fulfill the wants of sufferers, some challenges nonetheless hinder entry to the therapy for severely unwell sufferers. A definition of high quality requirements for FMT is of utmost significance resulting from security considerations across the transmission of multi-drug resistant organisms via FMT. Standardization of preparation processes greatest suited to protect functionalities of the intestinal ecosystem, growth of acceptable instruments to finely assess engraftment and documentation of compatibility between FMT drug and the recipient are main scientific challenges that this therapy modality wants to beat to be able to transfer from the bench to the bedside in keeping with Joël Doré, a skilled intestine microbial ecologist with 40 years’ expertise in each primary and translational developments in microbiome-based therapies. Within the present post-COVID-19 period, screening donors and recipients to make sure the efficacy and security of FMT can also be related.
Regardless of merchandise based mostly on FMT being among the many most superior microbiome-based remedies, the dearth of consistency in regulatory requirements and scientific tips derived from these requirements hinders its ready-to-use utility for sufferers. America has the longest historical past in taking the lead within the microbiome area. In 2013, the FDA positioned FMT for recurrent CDI as a therapeutic intervention that solely wants the knowledgeable consent of the affected person, whereas for different indications it’s thought of a drug and thus requires an investigational new drug utility to be made. In November 2022, a product composed of an outlined consortium of microorganisms was the first-ever microbiome therapeutic accepted by the FDA for stopping the recurrence of C. difficile an infection in adults after standard-of-care antibiotic therapy.
When making FMT available to severely unwell sufferers, Doré states that: “Manufacturing of the medicine shouldn’t be a limiting issue, however it nonetheless is in lots of international locations. The false notion that the educational/hospital constructions will be capable to fulfill the necessity might be at stake. It has to develop into the routine job of pharma firms to situation the FMT medicine and make them available.”
Australia has facilitated entry to fecal transplants for treating not solely C. difficile however different digestive situations. Final November, it grew to become the primary nation on the earth to obtain regulatory approval for fecal transplants as a organic drug for restoring the intestine microbiota within the administration of gastrointestinal issues.
In Europe, there’s a lengthy option to go earlier than advertising and marketing authorizations of FMT are granted. And despite the fact that the EU Innovation Community revealed a latest report with an replace of the regulatory state of affairs of FMT in Europe, the standard and security necessities of FMT is outlined by particular person international locations.
Most EU international locations have regulated FMT as a drug, whereas Finland regulates it as a therapeutic intervention and Italy as tissue. The UK additionally regulates FMT as a drug and Canada classifies it as a organic drug. When FMT is assessed as a pharmaceutical drug or organic product, it’s topic to extra stringent necessities and laws. As well as, the diploma of processing of the fecal materials or its indication additionally determines how FMT is regulated. Regardless of FMT being a widespread therapy in Europe, there’s a important underuse of FMTs (masking solely 10% of sufferers with recurrent CDI).
Elevating medical consciousness, adjusting regulatory frameworks for FMT and collaboration between scientists, clinicians and regulatory companies are advised as steps that may assist transfer the sector ahead.
In keeping with Doré: “By far the final consensus in Europe is to contemplate FMT merchandise as medicine, which might be the very best for the sufferers. A harmonized regulation for all international locations contemplating FMT merchandise as medicine could possibly be promoted. C. difficile an infection it’s the solely ecological illness for which FMT as an ecological therapy doesn’t appear extraordinarily demanding: the barrier operate that eliminates the pathogen could be restored with a preparation that doesn’t try and protect all functionalities of the intestinal ecosystem.”
Past regulatory points, one other problem is FMT utility in susceptible populations, reminiscent of kids. The pediatric inhabitants will likely be much less prone to profit from the therapy given the dearth of medical trials in kids and the constraints of accessible assessments to distinguish an infection from colonization in kids, which obstructs medical choices on the very best time to start out FMT remedy. As for every other drug, FMT might not be advisable for particular susceptible populations, till confirmed protected.
The way forward for fecal microbiota transplants
Whereas infectious ailments are the sector through which purposes of FMT are most generally researched, scientists have additionally began exploring its position for preserving the intestine microbiome broken after most cancers remedies and likewise for inflammatory bowel ailments, resulting from its immune-modulating properties. Examples of ailments through which FMT may develop into an accepted therapy choice sooner or later are irritable bowel syndrome, liver issues, decolonization of multidrug-resistant micro organism, metabolic issues, some neurological issues and at the same time as add-on to most cancers remedy.
In keeping with Doré: “The present scientific literature signifies that fecal microbiota switch will likely be far more potent than most if not any of the only pressure or small consortia examined in step with the probiotic idea. It’s going to nonetheless probably be add-on therapies within the many contexts for which customary of care is just not offering a treatment but additionally is barely environment friendly in a fraction of sufferers. Scientific proof has been constructing for ulcerative colitis, for most cancers remedy with immune checkpoint inhibitors or probably chimeric antigen receptor (CAR) T-cells, and probably for therapeutic dead-ends reminiscent of steroid-resistant acute graft versus host illness, acute-on-chronic liver failure or amyotrophic lateral sclerosis.”
Take-home messages
Whereas fecal microbiota transplants are generally used for medically refractory or recurrent difficile infections, questions stay about its effectiveness, security, regulation and greatest observe.
The US and Australia have accepted FMT for recurrent difficile an infection, whereas in Europe, regulation of the process hinders its widespread use, particularly amongst high-risk sufferers for whom the therapy is likely to be lifesaving.
Past gastrointestinal situations, immune-mediated ailments and oncology are areas of potential curiosity for fecal microbiota transplants and their utility in these contexts might happen within the close to future.
**********
References:
Kelly CP. Fecal microbiota transplantation – an outdated remedy comes of age. N Engl J Med. 2013; 368(5):474-475. doi: 10.1056/NEJMe1214816.
Gosálbez L. Microbiome Drug Improvement: 2022 in evaluate. Microbiome Instances. December 30, 2022. Accessible in: https://www.microbiometimes.com/microbiome-drug-development-2022-in-review/
Intestine Microbiota for Well being. A brand new research illustrates the dangers of administering fecal microbiota transplants to compromised sufferers. Schneider S. April 6, 2020. Accessible in: https://www.gutmicrobiotaforhealth.com/a-new-study-illustrates-the-risks-of-administering-fecal-microbiota-transplants-to-compromised-patients/
Kazemian N, Kao D, Pakpour S. Fecal microbiota transplantation throughout and post-COVID-19 pandemic. Int J Mol Sci. 2021; 22(6):3004. doi: 10.3390/ijms22063004.
U.S. Meals & Drug Administration. Science and regulation of stay microbiome-based merchandise used to stop, deal with, or treatment ailments in people. September 17, 2018. Accessible in: https://www.fda.gov/vaccines-blood-biologics/workshops-meetings-conferences-biologics/science-and-regulation-live-microbiome-based-products-used-prevent-treat-or-cure-diseases-humans#event-materials
BiomeBank. BiomeBank pronounces world first regulatory approval for donor derived microbiome drug. November 9, 2022. Accessible: https://www.biomebank.com/information/media-release/biomebank-announces-world-first-regulatory-approval-for-donor-derived-microbiome-drug/
European Medicines Company. Faecal Microbiota Transplantation. June 2022. Accessible in: https://www.ema.europa.eu/en/paperwork/report/faecal-microbiota-transplantation-eu-horizon-scanning-report_en.pdf
Hocking L, Ianiro G, Leong RW, et al. Faecal microbiota transplantation for recurrent C. difficile infections: challenges and enchancment alternatives for medical observe and healthcare programs. Aliment Pharmacol Ther. 2022. doi: 10.1111/apt.17309.
Merrick B, Allen L, M Zain NM, et al. Regulation, danger and security of faecal microbiota transplant. Infect Prev Pract. 2020; 2(3):100069. doi: 10.1016/j.infpip.2020.100069.
Hourigan SK, Nicholson MR, Kahn SA, et al. Updates and challenges in fecal microbiota transplantation for Clostridioides difficile an infection in kids. J Pediatr Gastroenterol Nutr. 2021; 73(4):430-432. doi: 10.1097/MPG.0000000000003229.
Ransom EM, Burnham CAD, Jones L, et al. Fecal microbiota transplantations: the place are we, the place are we going, and what’s the position of the medical laboratory? Clin Chem. 2020; 66(4):512-517. doi: 10.1093/clinchem/hvaa021.
Nationwide Institute for Well being and Care Excellence (NICE). Fecal microbiota transplant for recurrent Clostridioides difficile an infection. 31 August 2022. Accessible: https://www.good.org.uk/steerage/mtg71
Ianiro G, Segal JP, Mullish BH, et al. Fecal microbiota transplantation in gastrointestinal and extraintestinal issues. Future Microbiol. 2020; 15:1173-1183. doi: 10.2217/fmb-2020-0061.